Company Overview
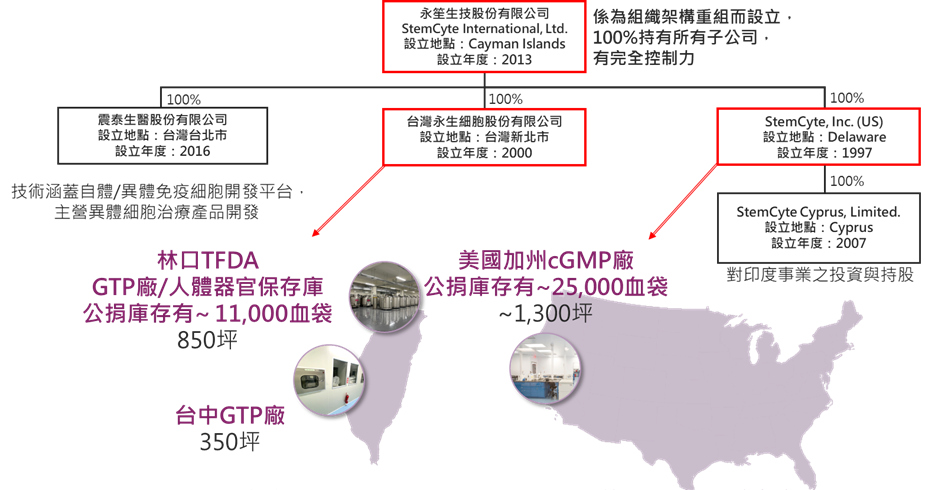
StemCyte International, Ltd. (“StemCyte-KY”) was established in 2013 as the holding company of the Group. Its wholly owned subsidiaries include StemCyte, Inc. in the United States (established in 1997) and StemCyte Taiwan (established in 2000), both of which have long specialized in cord blood and cell therapy. StemCyte-KY operates across Taiwan and the United States, with TFDA GTP facilities in Linkou and Taichung, a research center in Nangang, and a cGMP manufacturing facility in California, providing integrated capabilities in research, manufacturing, and global supply.
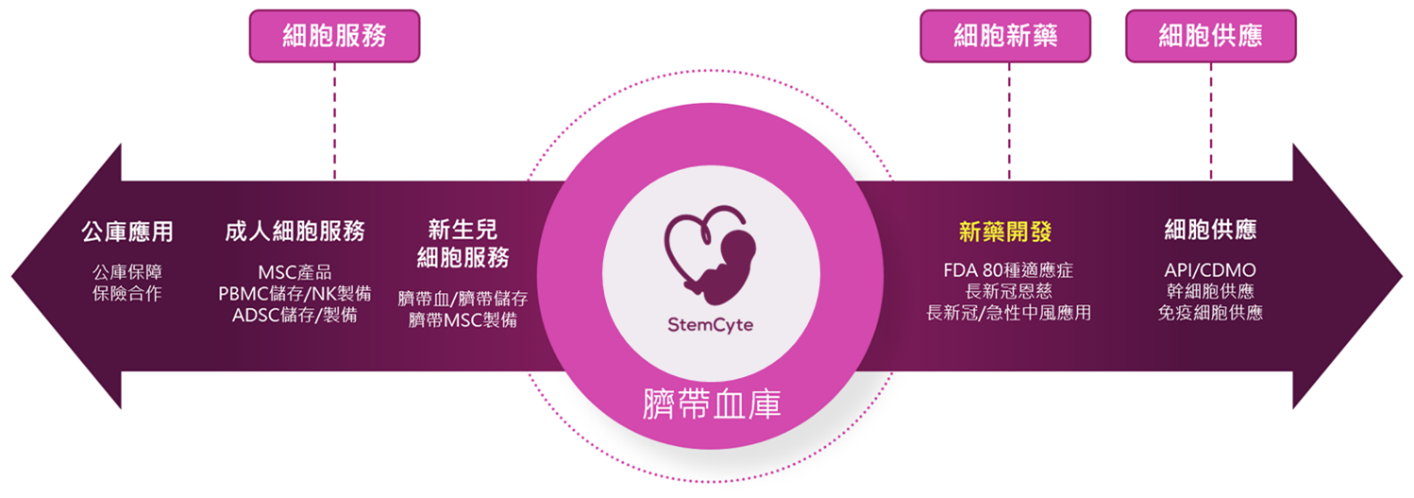
Leveraging its cord blood resources as the foundation, the Company integrates three core business segments—Cell Therapy, Cell Materials Supply, and Cell Services—to build an internationally competitive cell therapy platform.
Competitive advantage
StemCyte-KY operates Taiwan’s only public cord blood bank and one of the largest cord blood banks in the world. It currently maintains an inventory of more than 36,000 cord blood units, with over 2,300 transplants performed globally and partnerships with more than 350 medical centers worldwide. It is also the only public cord blood bank in Taiwan accredited by both AABB and FACT international quality standards.
With its scarce and valuable cell resource base, internationally recognized quality systems, and extensive clinical application track record, the Company has established a highly differentiated barrier to entry rarely seen in the regenerative medicine industry.
Core Product and Service- StemCyte's Cellular Three Arrows
Cell Therapy
The Company’s flagship product, REGENECYTE®, is the only cord blood-based cell therapy product in Taiwan to receive U.S. FDA Biologics License Application (BLA) approval. It is indicated for the treatment of nearly 80 hematologic and immune-related diseases.
In recent years, the Company has actively expanded into new indications, including Long COVID and acute ischemic stroke. Its Phase II clinical trial for Long COVID has successfully achieved topline results, and the program has received both Regenerative Medicine Advanced Therapy (RMAT) designation and approval for a fee-based Expanded Access Program (EAP) from the U.S. FDA, demonstrating the Company’s strengths in regulatory execution and commercial translation.

Cell Materials Supply
Leveraging its extensive cord blood inventory and internationally compliant manufacturing capabilities, StemCyte-KY provides cell sourcing and cell materials to medical institutions, research organizations, and industry partners. These services support transplantation therapies, clinical trials, and the growing needs of the regenerative medicine industry.
In addition, the Company is expanding into the cell therapy CDMO value chain by supplying API-grade cellular materials manufactured to U.S. FDA pharmaceutical standards, further strengthening its long-term growth momentum.
Cell Services
StemCyte-KY has introduced the world’s first Public (Cord Blood) Bank Access Model. By partnering with insurance providers, the Company integrates public cord blood matching services into healthcare reimbursement system. This model combines HLA typing databases with cell resource matching mechanisms to create a new service model for cell therapy.
This innovative approach not only expands service accessibility, but also transforms a traditionally one-time transaction model into a recurring and sustainable service revenue stream.
As the regenerative medicine industry continues to grow rapidly, StemCyte-KY will leverage its cord blood resource advantage as a foundation to further advance clinical applications in cell therapy, drive development of new indications, and actively expand overseas licensing partnerships and international market presence, with ongoing collaboration discussions across the United States, China, and Southeast Asia.
