Company Overview
Founded in 1989, TCM Biotech International Corp. (4169) is a profitable new drug development company with R&D capabilities, extensive distribution channels, and steadily growing revenue. Headquartered in Xizhi District, New Taipei City, the company has a molecular testing laboratory certified by LDTS and ISO 17025, GDP-approved warehousing for pharmaceuticals, medical devices, and cold chain for medical devices. Breaking away from the high risks and prolonged losses typically faced by early-stage biotech firms, TCM is backed by a steady cash flow from its distribution channels. Currently, its core R&D products have entered the late-stage harvest period, ready to make significant strides onto the international stage.
Strategy: Sustainable Cycle of R&D and Marketing
R&D in the biotech industry requires extensive time and massive funding. However, TCM has firmly established itself in both the capital markets and medical distribution channels through a dual-track strategy: "operations driving R&D, and R&D fueling growth. TCM's current revenue comes from the sales drugs and medical devices from our own licenses and licensed from agencies.
TCM’s sales channel including medical centers, regional & local hospitals and primary care clinics in Taiwan. This highly dense "medical distribution network" generates a stable and continuously growing cash flow, significantly reducing the financial uncertainties of new drug development. Most importantly, this extensive end-market medical network will serve as a powerful backing for TCM's proprietary new drugs and diagnostic products, enabling them to rapidly enter the market and convert into revenue upon launch.
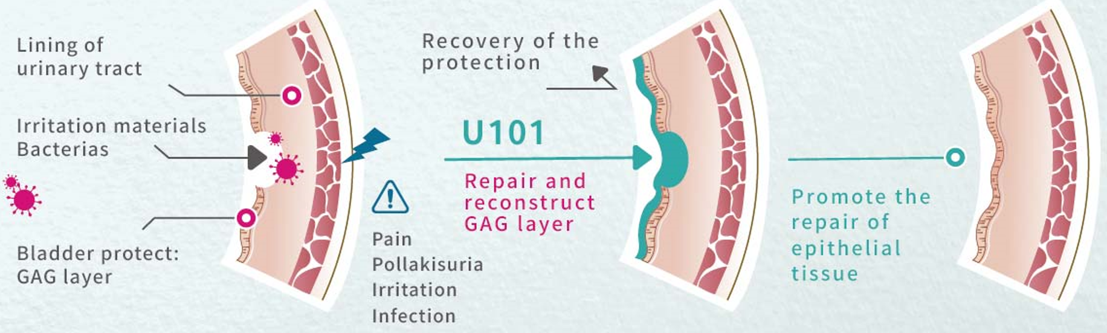
New Drug U101: The World's First Non-Antibiotic Therapy, Targeting the Massive Market of Recurrent Urinary Tract Infections
In terms of innovative R&D, TCM targets "unmet medical needs" in clinical settings. Its core new drug, U101, is the world's first non-antibiotic oral drug developed specifically for the prevention of recurrent urinary tract infections (rUTI).
Clinically, the recurrence rate of urinary tract infections is extremely high. Long-term reliance on low-dose antibiotics for prevention easily leads to antimicrobial resistance, while health supplements on the market show very limited evidence-based efficacy. With nearly 100 million potential rUTI patients worldwide, the market size is massive. U101 is being developed via the 505(b)(2) pathway, which effectively shortens the development timeline, while simultaneously securing the key active pharmaceutical ingredients (API). Currently, U101 has entered Phase III pivotal clinical trials in Taiwan. Following the upcoming interim analysis results, the company expects to submit a New Drug Application (NDA) in Taiwan and simultaneously submit Phase III IND in the US.
Precision Medicine HBV-Related Liver Cancer Diagnostic Platform "CatCHimera": FDA Breakthrough Device Designation Opens a New Chapter in Liver Cancer Recurrence Monitoring
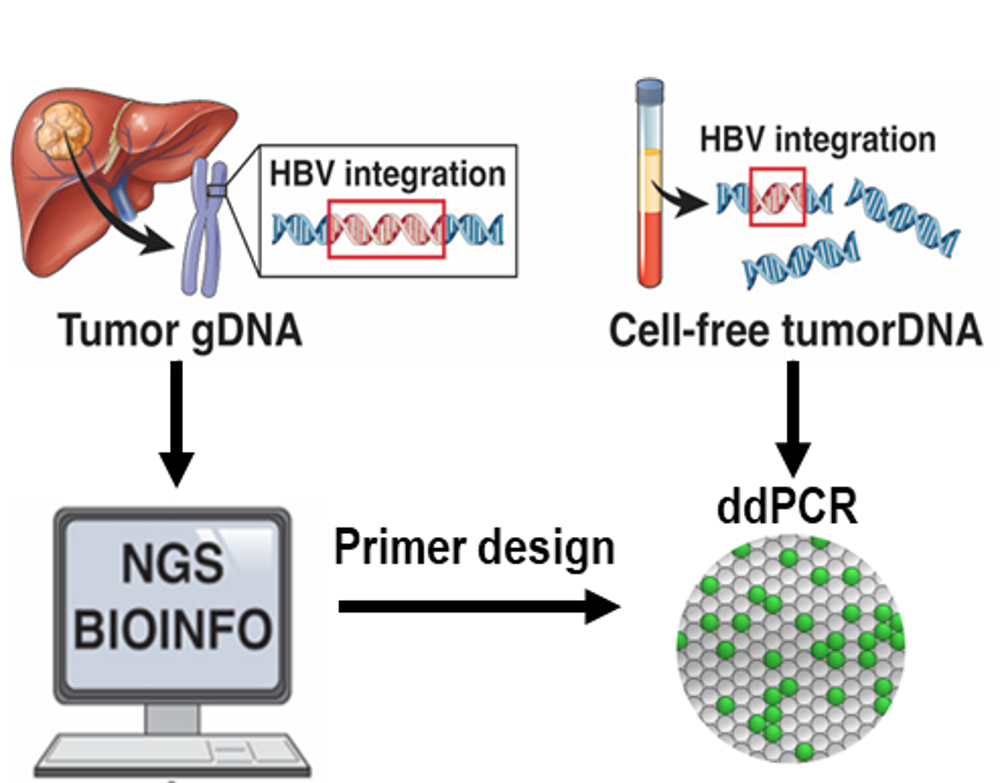
Addressing the critical pain point that post-operative recurrence rate for Hepatitis B-related liver cancer patients reach up to 50% within the first year, TCM has launched the CatCHimera liver cancer diagnostic platform.
Through blood-based liquid biopsies combining NGS and ddPCR technologies, this platform to identify tumor-specific HBV integration sites from resected HCC tissue. Published data show 98% pre-operative detection, 3 to 9 months lead time over CT imaging, 92% NPV for recurrence, and 95.8%/95.5% sensitivity/specificity when combined with AFP and PIVKA-II. CatCHimera has not only obtained LDTS certification from TFDA in Taiwan but has also been granted the "Breakthrough Device Designation" (BDD) by the US FDA. This prestigious designation will not only accelerate future regulatory reviews in the US but also lay a solid foundation for future technology licensing in China and global markets, targeting a massive monitoring market of 450,000 newly diagnosed liver cancer patients annually.
Future Outlook
Looking comprehensively at TCM’s advantages, its robust marketing channel ensures stable profitability that is impervious to economic fluctuations. Meanwhile, the high-potential products U101 and CatCHimera, both entering late-stage clinical and regulatory approval phases, strongly demonstrate the company's R&D capabilities.