EirGenix, Inc. (hereafter referred to as the Company) was established in December 2012 as a company focused on the development of biosimilar products. The Company provides Contract Development and Manufacturing Organization (CDMO) services, covering platforms such as cell line development, process development, analytical sciences, and protein characterization. The Company operates two PIC/S GMP-certified manufacturing facilities approved by the Taiwan Food and Drug Administration (TFDA), with capabilities in both mammalian cell and microbial production. With comprehensive quality control and assurance systems, the Company supplies clinical trial materials and commercial drug products to domestic and international clients.
Business Model and Core Technical Capabilities
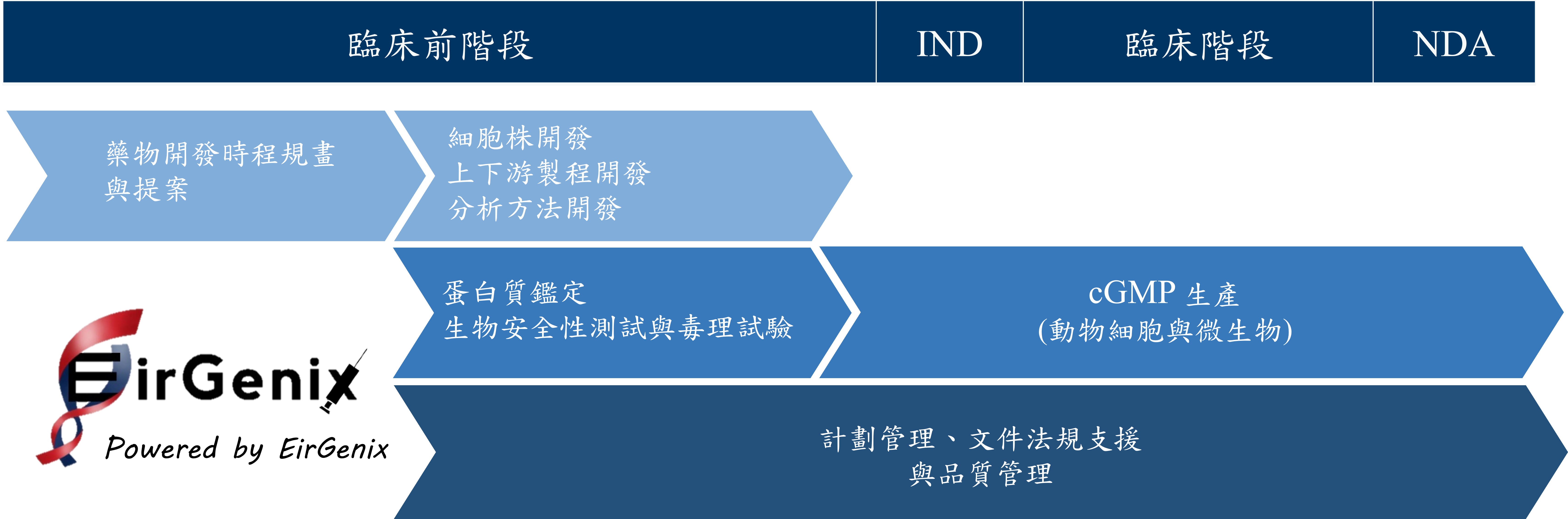
The Company adopts a two-pillar business model, combining Contract Development and Manufacturing Organization (CDMO) services with Product Development. By leveraging its cGMP-compliant manufacturing facilities and a highly skilled technical workforce, the Company is committed to developing high-quality, competitive biosimilar products. Its core competitiveness lies in its dual expertise in mammalian cell development and microbial strain fermentation—two key technologies. By integrating R&D, manufacturing, and analytical capabilities, and operating through a vertically integrated business model, the Company effectively controls product quality and reduces production costs.

Key Competitive Advantages
Building on a solid foundation in biologics development and manufacturing, the Company has continued to strengthen its competitive edge in the international market, with key advantages including:
- Possesses proprietary CHO-K1 cell lines and has full capability in cell line development.
- Process development, product analysis, and manufacturing are carried out by a professional team with international experience, ensuring technical quality.
- Possesses both mammalian cell line development and microbial strain fermentation technologies.
- The Zhubei facility has established a commercial-scale manufacturing facility compliant with international cGMP standards, offering robust quality control and production capabilities that meet global requirements.
- Possesses extensive regulatory experience in international clinical trial applications and marketing approvals.
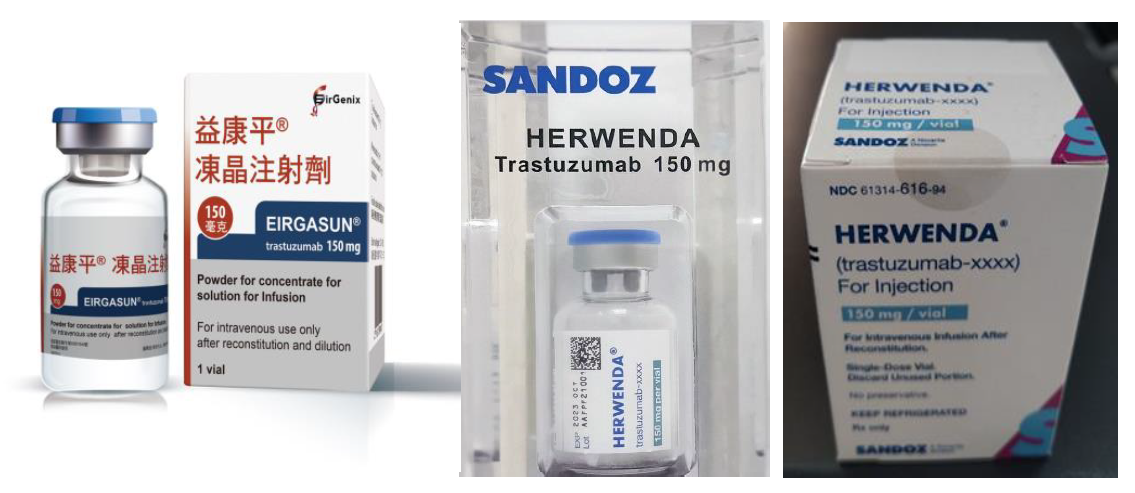
In addition, the Company’s first biosimilar product for breast cancer has obtained two drug approvals and has been launched in the Taiwan market. The Company has also formed a strategic alliance with Formosa Laboratories, Inc., combining the strengths of biologics and small-molecule drugs to develop antibody-drug conjugates (ADCs).

Future Prospects
EirGenix, Inc. is committed to becoming a key global base for biologics research and manufacturing. Guided by the vision of “Rooted in Taiwan, Reaching the Global Market,” the Company is actively expanding its business scope with a dual focus on biosimilar product development and CDMO services. Building on its existing technological and manufacturing foundations, EirGenix, Inc. continues to expand its product portfolio and production line layout, establishing a comprehensive biologics value chain that spans upstream research, midstream process development, and downstream manufacturing. At the same time, the Company is actively investing in the field of cell and gene therapy, offering innovative development and large-scale production of DNA plasmids for partnered gene delivery systems.
Since its inception, EirGenix, Inc. has focused on providing high-quality and cost-effective CDMO services for biologics, while also developing high-quality and affordable biosimilar products. Looking ahead to the medium- and long-term, the Company will continue to expand its international services and partnerships in the biosimilar sector and progressively move into the development of niche biologics and innovative biologic drugs. Committed to advancing human health and enhancing quality of life, EirGenix is steadily progressing toward its goal of becoming a globally influential biopharmaceutical company.
